Amount of substance
1. Vypočítajte počet molekúl oxidu dusnatého v nádobe, ktorá obsahuje 4,5 mol tohto plynu.
Riešenie:
Vychádzame z rovnice pre výpočet látkového množstva:
N(NO) = n(NO).NA = 4,5 mol. 6,022.1023 = 2,7.1024
V nádobe sa nachádza 2,7 . 1024 molekúl NO.
2.
Aké látkové množstvo predstavuje 1,806. 1027 atómov uhlíka?
Riešenie:
Použijeme vzťah pre látkové množstvo.
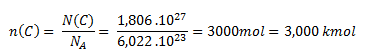
Látkové množstvo uhlíka obsahujúce 1,806. 1027 atómov, je 3 kmol.
3. Koľko molekúl je v nádobe obsahujúcej 2,5 mol oxidu uhličitého?
Riešenie:
Použijeme obdobný postup ako v príklade 2.
1,5 . 1024 molekúl.
4. Vypočítajte látkové množstvo:
- a) 2,58 . 1027 atómov hliníka
- b) 5,55 . 1019 sodných katiónov
- c) 8,68 . 1022 chloridových aniónov
- d) 1,75 . 1025 molekúl bieleho fosforu (P4)
Riešenie:
Použijeme rovnaký postup riešenia ako v príklade 2.
- a) 4,28 kmol
- b) 9,22 . 10-2 mmol
- c) 0,144 mol
- d) 29,1 mol
5. Náboj elektrónu je 1,6 . 10-19C. Aký náboj má 1 mol elektrónov (Faradayov náboj)?
Riešenie:
Podľa Avogardovej konštanty sa v jednom mole nachádza 6,022.1023 častíc. Každá častica, v našom prípade elektrón, nesie náboj 1,6 . 10-19 C. Jeden mol elektrónv teda nesie náboj 1,6 . 10-19. 6,022. 1023 čo sa rovná 96352 C.
6.
Koľko atómov striebra obsahuje 1 cm3 tohto kovu, ktorý má hustotu ς= 10,5 . 103 kg.m-3?
Riešenie:
Z hustoty a objemu vypočítame hmotnosť striebra:
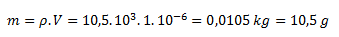
Mólová hmotnosť Ag je 107,868 g/mol, pre látkové množstvo striebra teda platí:
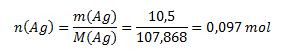
a pre počet častíc:
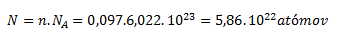
1 cm3 striebra obsahuje 5,86 . 1022 atómov.
7.
Aké látkové množstvo H2O je v jednom litri vody?
Riešenie:
Hustota vody je 1 g/cm3 , preto liter vody váži 1 kg, resp. 1000g. Mólová hmotnosť vody je 18 g/mol, pre látkové množstvo teda platí:
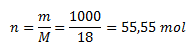
Liter vody obsahuje 55,5 mol molekúl H2O.
8.
Aké látkové množstvo sodíka je v 10 g NaCl? Koľko atómov chlóru pripadá v tejto zlúčenine na dané látkové množstvo sodíka?
Riešenie:
Postup riešenia je obdobný ako v príklade 6.
0,17 mol Na; 1,03 . 1023 atómov Cl
9. Akému látkovému množstvu zodpovedá 1g H2SO4 a 200 g CH4?
Riešenie:
Postup riešenia obdobný ako v príklade 6.
0,01 mol; 12,47 mol
10. Aké látkové množstvo hliníka je v kocke s hranou 10 cm? (použite údaje z chemických tabuliek)
Riešenie:
Postup riešenia ako v príklade 6.
100,07 mol