Work of gas
1. Explain the work of a gas and the cyclic process.
Solution:
Hot gas or steam can perform work. Work of a gas at constant pressure:
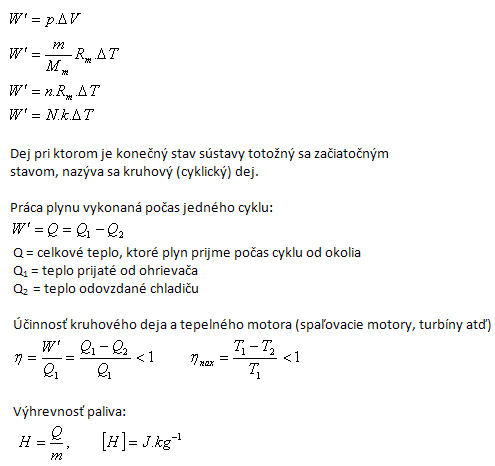
2.
Calculate the work of steam during one piston stroke if the steam pressure on the piston is 176.104 Pa and the piston area is 700 cm2. The piston stroke is 55 cm.
Solution:
Analysis:
p = 176.104 Pa, S = 700.10-4 m2, h = 55.10-2 m,
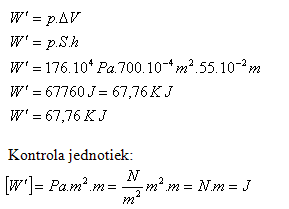
The work of steam during one piston stroke is W = 67.76 kJ
3.
Oxygen O2 (Mm = 32.10-3 kg·mol-1) has a mass of 4 kg and a temperature of 0oC. How much will its temperature increase in an isobaric process if the gas performs work of 10.4 kJ?
Solution:
Analysis:
m = 4 kg, Mm = 32.10-3 kg·mol-1, W = 10.4·103 J, Rm = 8.314 J·kg-1·K-1
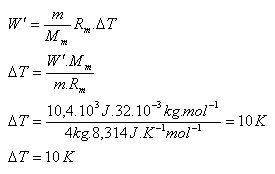
The gas temperature increases by ΔT = 10 K.
4. What is the mass of fuel consumed by airplane engines on a flight of 500 km if the average speed is 250 km·h-1, the average engine power is 2 MW, engine efficiency is 25%, and the fuel calorific value is 45 MJ·kg-1?
Solution:
Analysis:
s = 500 km = 5·105 m, v = 250 km·h-1 = 69.44 m·s-1,
P = 2·106 W, H = 45·106 J·kg-1, η = 0.25 m = ?
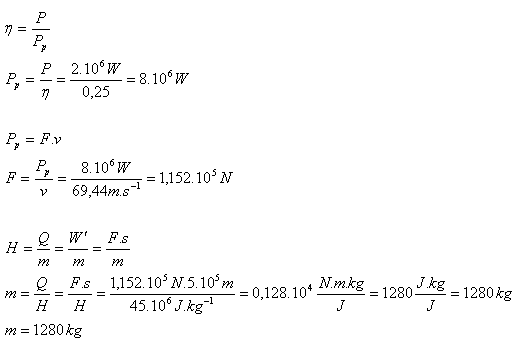
The airplane engines consume 1280 kg of fuel.
5.
A combustion engine receives heat of 1·104 J from a heater at 900oC. It transfers 75% of this heat to a cooler at 150oC. What is the engine efficiency? What is the maximum efficiency of an engine operating between these temperatures?
Solution:
Analysis:
T1 = 1173.15 K, T2 = 423.15 K, Q1 = 1·104 J, Q2 = 0.75·104 J
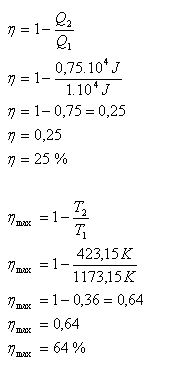
The engine efficiency is η = 25%, maximum efficiency η max = 64%.
6.
What is the “second law of thermodynamics”?
Solution:
It is impossible to construct a periodically operating heat engine that would convert all the heat received from a heater into useful work.
It is impossible for heat to spontaneously transfer from a colder body to a hotter body.
The “second law of thermodynamics” states that it is impossible to construct a “perpetual motion machine of the 2nd kind,” i.e., a heat engine that operates without losses and with 100% efficiency.
7. Complete the short test:
- 1.) The molar mass of carbon dioxide CO2 is: (12C, 16O)
A.) 44·10-3 kg·mol-1 B.) 55·10-3 kg·mol-1 C.) 33·10-3 kg·mol-1
- 2.) Which quantity does not belong among the basic state variables?
A.) temperature B.) volume C.) time
- 3.) An ideal gas has volume V at 27oC. At what temperature does it have volume 0.75V?
A.) –31oC B.) 48oC C.) –48oC
- 4.) An ideal gas has a pressure of 5 MPa at 0oC. Determine its pressure at –50oC.
A.) 3.04 MPa B.) 4.08 MPa C.) 5.03 MPa
- 5.) What is the calorific value of fuel if burning 200 kg of fuel releases 620·107 J of heat?
A.) 3.1·107 J·kg-1 B.) 5.4·106 J·kg-1 C.) 6.9·105 J·kg-1
- 6.) What is the maximum efficiency of a steam turbine if the heater temperature is 530oC and the cooler temperature is 50oC?
A.) 60 % B.) 65 % C.) 70 %
- 7.) What work does an ideal gas of 1 mol perform if its temperature increases by 1 K at constant pressure?
A.) 1.38 J B.) 8.314 J C.) 6.022 J
Solution:
1A, 2C, 3C, 4B, 5A, 6A, 7B